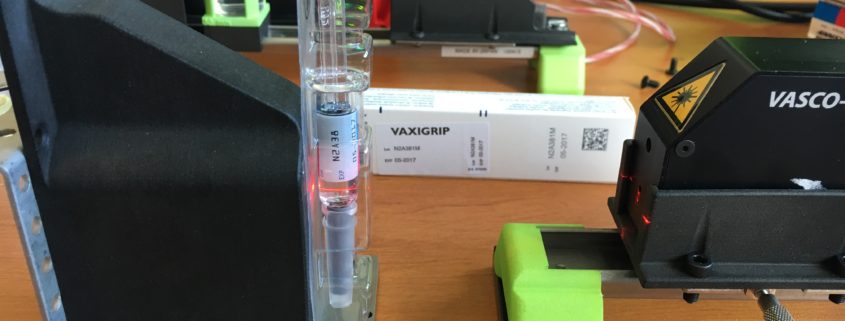
Monitoring protein aggregation in injectable vaccines syringes
Monitoring protein aggregation in injectable vaccines syringes: a new direct & contact-less technique
David Jacob CTO
Keywords: vaccines, protein, in situ measurement, aggregation
AN01-VASCO Kin – monitoring Protein aggregation in injectable
Aggregation of proteins and active principle ingredients (API) in injectable biopharmaceutical products remains a major concern impacting the stability and usability of a product. Indeed, Protein aggregation can occur during all stages of the lifetime of a protein therapeutic, including expression, refolding, purification,
sterilization, shipping, storage, and delivery processes [1, 2]. The mechanism of protein
aggregation is still not well understood; but it is known that certain manufacturing stages
like formulation composition, presence of microbial or vial contaminants during cell
culture, and storage influence the risk of chemical degradation, which increases the risk
of physical degradation and the formation of aggregates. In particular, it has been shown
that the storage container environment plays a role like in prefilled syringes for example
where leaking silicone oil from the rubber stopper, and glass delamination can induce
aggregates. In a context of more and more stringent international health regulations
about the control of biopharmaceutical products, the in-situ monitoring of the denaturation and degradation process of therapeutic proteins during production and storage can be a key competitive advantage for manufacturers and researchers